|
Ethnobotanical Leaflets 9: 15- 23. 2005
Promising Phytochemicals from Indian Medicinal Plants
Dr. Amrit Pal Singh MD (Alternative Medicine), Medical Executive, Ind-Swift Ltd, Chandigarh (India)
Address for correspondence: Amrit Pal Singh House No. 2101, Phase-7, Mohali-160062 Email:
Issued 16 May 2005
Abstract The medicinal plants find application in pharmaceutical, cosmetic, agricultural and food industry. The use of the medicinal herbs for curing disease has been documented in history of all civilizations. With the onset of research, it was concluded that plants contain active principles, which are responsible, for curative action of the herbs. Ayurvedic drugs are used in crude forms like expressed juice, powder, decoction or infusion. Ancient healers, developed formulations based on medicinal herbs, were probably not aware about the chemical composition of the herbs. But the advancement they made despite non-availability of scientific procedures is astonishing. Scientific research has proved the utility of time tested remedies. The article reviews work done on isolated constituents of rare Indian medicinal plants. Key Words: Ayurveda/ alkaloids/glycosides/medicinal herbs.
Introduction Medicinal herbs are significant source of synthetic and herbal drugs. In the commercial market, medicinal herbs are used as raw drugs, extracts or tinctures. Isolated active constituents are used for applied research. For the last few decades, phytochemistry (study of plants) has been making rapid progress and herbal products are becoming popular.
Ayurveda, the ancient healing system of India, flourished in the Vedic era in India. According to historical facts, the classical texts of Ayurveda, Charaka Samhita and Sushruta Samhita were written around 1000B.C.The Ayurvedic Materia Medica includes 600 medicinal plants along with therapeutics. Herbs like turmeric, fenugreek, ginger, garlic and holy basil are integral part of Ayurvedic formulations. The formulations incorporate single herb or more than two herbs (poly-herbal formulations).
Medicinal herb is a considered to be a chemical factory as it contains multitude of chemical compounds like alkaloids, glycosides, saponins, resins, oleoresins, sesquiterpene lactones and oils (essential and fixed). Today there is growing interest in chemical composition of plant based medicines. Several bioactive constituents have been isolated and studied for pharmacological activity.
Clausena anisata Clausenol (carbazole alkaloid) isolated from Clausena anisata has shown antibacterial activity. The phytochemical was found to be active against bacteria and fungi.
Ceropegia juncea Cerpegin (a furopyridine alkaloid) from Ceropegia juncea has shown analgesic effect against acetic acid induced writhing in mice.
Sarcocca pruniformis Saracocine reported from Sarcocca pruniformis has not been investigated for pharmacological activity.
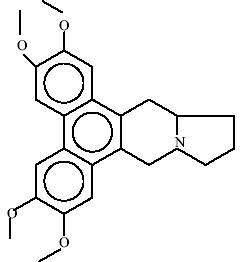
Tylophora asthamatica Experiments conducted with Tylpohorine (has phenanthroindalizidine framework). in various animal models have shown significant anti-inflammatory, anti-anaphylactic and anti-spasmodic activites. Pre-treatment with Tylophora asthmatica prevented bronchospasm induced by Freund’s adjuvant and bovine albumin in rats. The same researchers observed that the plant extract produced muscle relaxant activity, antagonism of smooth muscle stimulants and immunosuppressive effects in different species.
Structure of Tylophorine
Swertia chirata
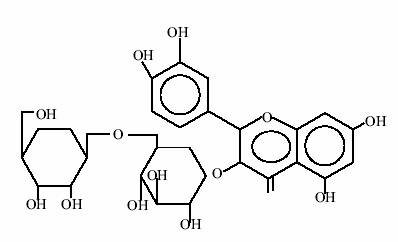
Amarogentin, bitter principle of Swertia chirata (chirata) has anti-leishmanial activity.27
Structure of Amarogentin
Swerchirin, xanthone from Swertia chirayita (Chirata) has antidiabetic activity. Researchers compared the effects of mode of action of three different hypoglycemic agents; centipiperalon, tolbutamide and swerchinin in normal as well as diabetic rats. Except in rats with severe pancreatic damage, swerchinin showed better glucose lowering effect compared to tolbutamide.
Hibiscus vitofolius
Gossypin from Hibiscus vitofolius (Japa) has anti-nociceptive activity similar to opium alkaloids and involving multineurotransmiter systems. . It acts through cholinergic and GABAergic pathways. It seems to have potential analgesic activity with free from tolerance and dependence.
Sophora japonica
Rutin, a bioflavonoid isolated from Sophora japonica reduced the infract size an prevented the loss of the R wave in anaesthetized rats subjected to coronary artery ligation.The drug however not effected the heart rate and systolic blood pressure. The cardio protective action of the drug is assumed due to antioxidant activity of rutin.
Structure of Rutin
Abies pindrow
Extracts of Abies pindrow demonstrated anti-inflammatory activity in various models of inflammation such as carrageenin induced paw oedema, granuloma pouch and Freund’s adjuvant arthritis. Chemical analysis indicated presence of glycosides, terpenoids and flavonoids. The flavonoids are terpenoids are effective in acute inflammation and glycosides are effective in chronic inflammation.
Thevetia nerifolia
Thevetin, a glycoside from Thevetia nerifolia (Karvira) has digitalis like activity.36
Plumbago rosea
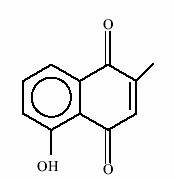
In animal studies, plumbagin, napthoquinone from Plumbago rosea (Chitraka) has shown anti tumour activity. The antitumour and radimodifying properties of plumbagin were tested on mouse Ehrlich ascites carcinoma. Plumbagin produced inhibition of exponentially growing tumours. When radiation was combined with plumbagin, mouse survival was increased by 120 days. However mode of action of anti cancer activity of plumbagin remains unclear.
Structure of Plumbagin
Curcuma longa
Curcumin from Curcuma longa (Haridra) has antioxidant, anti-inflammatory, anti cancer, and hepatoprotective. The pharmacological activities of cucrcumnoids are due to unique molecular structure.
Aegle marmelos
Luvangetin, pyranocoumarin from Aegle marmelos (Bilva), has shown potent gastroptroective activity. It gave significant protection against pylorus ligation and aspirin induce gastric ulcers in rats and cold restraint-induced gastric ulcers in rats and guinea pigs.
Nelumbo nucifera
Methanolic extract of Nelumbo nucifera rhizome and betulinic acid, a triterpene isolated from the plant demonstrated significant anti-inflammatory activity when tested in carrageenin and 5-hydroxytryptamine induced paw edema. The activity was comparable to betamethasone and phenylbutazone.
Structure of Betulinic acid
Crateva nurvala
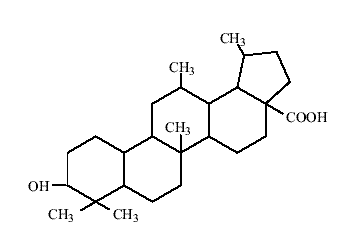
Lupeol and other chemical constituents from Crateva nurvala possessed significant anti-hyperxaluric and anti-hypercalcuric activity when tested in rats against hydroxyproline induced hyperoxaluria and calciuria.
Rubia cirdifolia
Rubidianin, an anthraquinone isolated from alcoholic extract of Rubia cordifolia has demonstrated significant antioxidant activity as it prevented lipid peroxidation induced by ferrous sulphate and t-butylhydroperoxide. The drug depicted activity in dose-dependent manner. The anti-oxidant activity of rubidianin was found to be better than mannitol, vitamin e and p-benzoquinone.
Boswellia serrata
Boswellic acid, from Boswellia serrata (Salai guggul) has anti-inflammatory and immunomodulator. Boswellia serrata is widely prescribed in the treatment of osteoarthritis, rheumatoid arthritis and bronchial asthma.
Elephantus scaber
Elephantopin from Elephantus scaber (Gojihva) has activity against the P388A mouse leukemia. Recently leaves of Elephantopus scaber have been investigated against Dalton’s ascitic lymphoma in animal models.
Cedrus deodara
Himachalol from Cedrus deodara (Deodara) has shown potent anti-allergic activity.
Nardostachys jatamansi
Jatamansin from Nardostachys jatamansi (Jatamansi) has shown tranquillizing activity.
Butea monosperma
Palasonin from Butea monosperma inhibited glucose uptake and depleted the glycogen content in Ascaridia galli.
Eclipta alba
Wedelolactone and demethylwedelolactone from Eclipta alba (Bhringraja) have potent trypsin inhibitory effect.
Conclusion: With onset of scientific research in Ayurvedic system of medicine, it is becoming clearer that the medicinal herbs have a potential in today’s synthetic era, as numbers of medicines are becoming resistant. According to one estimate only 20% of the plant flora has been studied and 60%of synthetic medicines owe their origin to plants. Ancient knowledge coupled with scientific principles can come to the forefront and provide us with powerful remedies to eradicate the diseases. References:
1. Fluckiger, Archiv of Pharm .B.191, P.196.
2. Groves, Pha.Jour and Trans.1873-74, P.293.
3. Rep. Chem .Exam for Bengal, 1885.
4. Rajani, M and K. Pundarikakshudu (1996).A note on seasonal variation of alkaloids in Adhatoda vasica Nees. International Journal of Pharmacognosy 34(4): 308-309. {a}Dep. Pharmacognosy, J.J. De Chane Lab. Pvt. Ltd., Residency Road, Hyderabad 500 001, India.
5. Shah et. al., J. Ass Physicians India, 1965, 13, 845.
6. Chakeaborty A, Chowdhury BK, Bhattacharya P. Clausenol and Clausenine –two carbazole alkaloids from Clausena anisata. Phytochemistry 1995; 40:295-8.
7. Herr Warneke. Berichte, XIX. 60.
8. Stajnow.Amer.Journ.Pharm., 1890, P 334.
9. Gibson. Pharmaceutical journal. xii., p.422.
10. O.Fisher and E.Tacaber.Ber.d.Chem.Gesselsch.1885, 400-406.
11. Kapoor LD. CRC Handbook of Ayurvedic Medicinal Plants. Boca Raton: CRC Press, 1990.
12. Archiv. der Pharmacie, Vol. XI .,p.19.
13. F.A.Ringer and E.Brooke. Amer.Jour.Pharm.1892. 14. Pharmacographia, 2nd Ed., p.291 . 15. Shankaracharya NB, et al. Characterisation of chemical constituents of Indian long pepper (Piper longum L). Journal of Food Science & Technology-Mysore 1997; 34:73-5.
16. Bai YF, Xu H. Protective action of piperine against experimental gastric ulcer. Acta Pharmacologica Sinica 2000; 21:357-59.
17. Sivarajan VV. Ayurvedic Drugs and their Plant Sources. Lebanon, New Hampshire: International Science Publisher, 1994.
18. Chopra RN, Chopra IC. Handa KK and Kapur LD. lndigeneous Drugs of India, 2nd ED, M/S U. N. Dhar &Sons Ltd. Calcutta,
19. J.M.Kohli, A. Zaman and A.R. Kidwai. Alkaloid of Sarcococca pruniformis. Phytochemistry 1971, Vol.10, pp. 442-445.
20. Watt, Dict. of Chem.viii.1807.
21. E.Schimdt and Schutte.Apoth.Zt.,1890,501.
22. Sen, R., D. C. Pal, et al. (1983). Traditional uses and ethnobotany of "Kuchila" (Strychnos nux vomica). Journal of Economic and Taxonomic Botany 4(2): 575-578.
23. Warden. Ind.Med.Gaz., Oct.1880.
24. E.Jahns. Bericht, 18, 2518-2523.
25. Gopalakrishnan C, Shankaranarayan D, Kameswaran L, et al. Pharmacological investigations of tylophorine, the major alkaloid of Tylophora indica. Indian J Med Res 1979; 69:513–20.
26. Thopra, R.K., S.G.Agarwal, et al. (1996). Two pyrroloquinazolines from Adhatoda vasica. Phytochemistry Oxford 42(5): 1485-1488. {a} Regional Res. Lab., Jammu Tawi-India.
27. Ray S, Majumdar HK, Chakarvarty AK, et al. Amarogentin, a naturally occurring secoiridoid glycoside and a newly recognized inhibitor of topoisomerase 1 from Leishmania donovani. J.Nat Prod 1996; 59:27-9.
28. Trivedi NP, Rawal UM. Hepatoprotective and antioxidant property of Andrographis paniculata(Nees) in BHC induced liver damage in mice. Indian J Exp Biol 200139 (1):41-46.
29. Rana AC, Avadhoot Y. Hepatoprotective effects of Andrographis paniculata against carbon tetra-chloride induced liver damage. Arch Pharm Res 1991:14 (1):93-95.
30. Isnard. Journ. de Phar. et de Chim., Aug. 1st, 1886.
31. Saraswat B, Visen PK, Patnaik GK, Dhawan BN. (997) Protective effect of picroliv, active constituent of Picrorhiza kurrooa, against oxytetracycline induced hepatic damage. ICMR Centre for Advanced Pharmacological Research on Traditional Remedies, Central Drug Research Institute, Lucknow, India. Indian J Exp Biol. Dec; 35(12): 1302-5.
32. Santra A, Das S, Maity A, Rao SB, Mazumder DN. (1998) Prevention of carbon tetrachloride-induced hepatic injury in mice by Picrorhiza kurrooa. Department of Gastroenterology, Institute of Post Graduate Medical Education and Research, Calcutta. Indian J Gastroenterol. Jan; 17(1): 6-9.
33. Satya Prakash and Asif Zaman. Flavonoids of Tinospora malabarica. Phytochemistry, Vol.21 No.12, pp.2992-2993, 1982.
34. Ramaswamy S, Vishwanathan S. Influence of gossypin on the development of acute tolerance to morphine induced antinociception. Indian J Expt Biol 1997; 35:413-4.
35. Saxena AM, Murthy PS, Mukherjee SK. Mode of action of three structurally different hypoglycemic agents: a comparative study. Indian J Exp Biol 1996; 34:406-9.
36. Arora, R.B.and Rangaswamy, S. (1972 A): Peruvoside and other cardiotonic glycosides, No.1:1-109, Thompson Press (India) Ltd., New Delhi.
37. Antihyperglycemic effects of gymnemic acid IV, a compound derived from Gymnema sylvestre leaves in streptozotocin-diabetic mice. Sugihara Y, Nojima H, Matsuda H, Murakami T, Yoshikawa M, Kimura I. Department of Chemical Pharmacology, Toyama Medical and Pharmaceutical University, Sugitani, Japan.
38. Uma Devi, F.E.Solmon and A.C. Sharda. Plumbagin, A Plant Napthoquinone with Antitumour and Radimodifying Properties. Pharmaceutical Biology 1999, Vol.37, No.3, pp. 231-236.
39. Arora, R.B. et al. (1971) Anti-inflammatory studies on Curcuma longa, L. Indian J.Med. Res. 59, 1289.
40. Majeed,M. et al.(1995) Curcuminoids : Antioxidant Phytonutrients. NutriScience Publishers Inc., Piscataway, New Jersy.
41. Mukundan, MA. et al. (1992).Effect of turmeric and curcumin on BP-DNA adducts, Carcinogensis 14,493.
42. Goel RK, Maiti RN, Manickam M, Ray AB. Antiulcer activity of naturally occurring pyrano-coumarin and iso-coumarins and their effect on prostanoid synthesis using human colonic mucosa. Indian J Exp Biol 1997; 35:1080-3.
43. Safayhi I, Mack T, Sabieraj J, Anazodo M I, Subramaniam L R, Ammon HPT. Boswellic acids: novel, specific, non redox inhibitors of 5-lipoxygenase.J.Pharmacol Exp Ther 1992, 261: 1143-1146.
44. Gupta I, Gupta V, Parihar A, Gupta S, Ludtke R, Safayhi H, Ammon HPT. Effects of Boswellia serrata gum resin in patients with bronchial asthma: Results of a double-blind, placebo–controlled, 6 week study. Eur J Med Rs 1998; 3: 511-514.
45. Sharma ML, Kaul A, Khajuria A, et al. Immunomodulatory activity of Boswellic acids (Pentacyclic Triterpene Acids) from Boswellia serrata. Phytotherapy Res 1996; 10:107-112.
46. Satyavati, GV (1991) Guggulipid: A promising hypolipidemic form gum guggul (Commiphora wightii). Economic and Traditional Medicine, 47-80.
47. Duwiejua M, Zeitlin I J, Waterman P. G, Chapman J, Mhango GJ and Proven G J. Anti-inflammatory Activity of Resins form some Species of the Plant Family Burseraceae. Planta Medica. 59 (1):12-6, 1999 Feb.
48. Bonte F et al. Influence of asiatic acid, madecassic acid, and asiaticoside on human collagen I synthesis. Planta medica, 1994, 60:133–135.
49. Mukherjee GD, Dey CD. Clinical trial on Brahmi. I. Journal of Experimental Medical Sciences 1966; 10:5-11.
50. Bhattacharya SK, Ghosal S. Anxiolytic activity of a standardized extract of Bacopa monniera - an experimental study. Phytomedicine 1998; 5: 77-82.
51. Stough C, Lloyd J, Clarke J, Downey LA, Hutchison CW, Rodgers T, Nathan PJ. The chronic effects of an extract of Bacopa monniera on cognitive function in healthy human subjects. Neuropsychology Laboratory, School of Biophysical Science and Electrical Engineering, Victoria, Australia.
52. Dar A, Channa S. Calcium antagonistic activity of Bacopa monniera on vascular and intestinal smooth muscles of rabbit and guinea pig. Pharmacology Section, HEJ Research Institute of Chemistry, University of Karachi, Pakistan. J Ethnopharmacol 1999 Aug; 66(2): 167-74.
53. SK, Bhattacharya A, Kumar A, Ghosal S. Antioxidant activity of Bacopa monniera in rat frontal cortex, striatum and hippocampus. Department of Pharmacology, Institute of Medical Sciences, Banaras Hindu University, Varanasi - 221005, India. Phytother Res 2000 May; 14(3): 174-9.
54. M.B.Gupta; R.Nath; N. Srivastava; K.Shanker; K Kishor and K.P.Bhargava. Anti-inflammatory and anti-pyretic activities of ß-sitosterol. Planta Medica, Vol.39, pp.157-163, 1980.
55. Asthana, R and Raina, M.K., (1989). Pharmacology of Withania somnifera-A review. Ind .Drugs; 26; 1-7.
56. Subhir, S et al., Pharmacological investigation on the leaves of Withania somnifera. Ind. J. Pharmacol; 17: 4-52.
57. Rajkapoor B; Jayakar B; Anadan R. Antitumor activity of Elephantpous scaber linn against dalton’s ascitis lymphoma. Indian Journal of Pharmaceutical Sciences.2002 Jan-Feb; 64(1): 71-3.
58. Gupta PP, Kulshrestha DK, Patnaik GK. Antiallergic activity of Cedrus deodara. J.Med Aromatic Plant Sci 1997; 19:1007-8.
59. Amatya G. Sthapit VM. A note on Nardostachys jatamansi. Journal of Herbs,Spices and Medicinal Plants.1994, 2: 2, 39-47.
60. Kumar D, Mishra SK, Tandan SK, Tripathi HC. Possible mechanism of antihelmintic action of palasonin on Ascaridia galli. Indian J Pharmacol 1995; 27: 161-8.
61. Syed SD, Deepak M, Yogisha S, Chandrashekar AP, Muddarachappa KA, D’Souza P, Agarwal A, Venkataraman BV. Trypsin inhibitory effect of wedelolactone and demethylwedelolactone. Phytother Res. 2003 Apr; 17 (4): 420-1.
|