|
Ethnobotanical Leaflets 13: 1197-1204. 2009.
A Comparative Study of the Antibacterial Activity of Piliostigma reticulatum Bark Extract with Some Antibiotics
1S. Awe and 2P.F. Omojasola
1Department of Biological Sciences, Ajayi Crowther University, Oyo, Nigeria E-mail: 2Department of Microbiology, University of Ilorin, Ilorin, Nigeria E-mail:
Issued 01 September 2009
Abstract
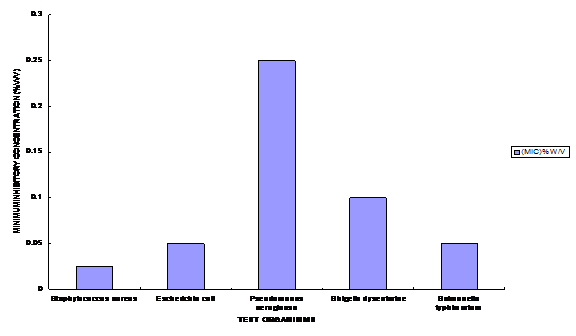
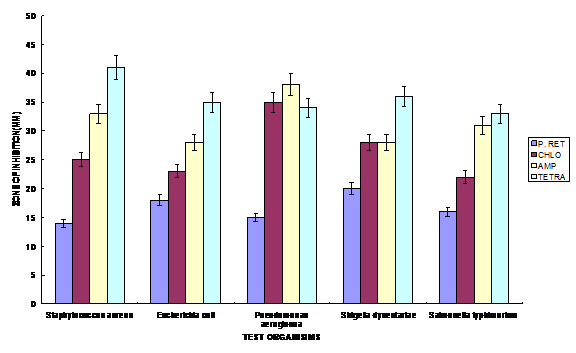
Aqueous and ethanolic extracts of Piliostigma reticulatum, a herb widely used for the treatment of diarrhea in southwest Nigeria, was challenged with clinical isolates of Escherichia coli, Shigella dysenteriae, Salmonella typhimurium, Staphylococcus aureus and Pseudomonas aeruginosa. The ethanol extract exhibited the highest activity against the test organisms with zones of inhibition ranging from 16.0 – 20.0 mm, followed by the hot aqueous extract and the cold aqueous extracts, with zones of inhibition ranging between 8.0 – 10.0 mm and 4.0 – 7.0 mm respectively. The Minimum Inhibitory Concentration (MIC) ranged between 0.025 – 0.25% w/v and photochemical analysis showed that the plant possesses saponins, tanins, phenolics, phlobatinins and glycosides. A comparative analysis of the extract with some conventional antibiotics namely; Chloramphenicol, Ampicillin and Tetracycline showed that there was significant difference in the antibacterial activity of the bark extract and the antibiotic standard (P<0.05). Key words: Antimicrobial activity, Minimum Inhibitory Concentration, Antibiotics, Phytochemical and Piliostgma reticulatum. Introduction Herbal medicine has been shown to be effective and about 60% of rural populations depend on it for their primary health care (WHO, 1988; Akinyemi et al., 2000). This could be attributed to affordability, accessibility, in the economic sense and socially, an uneven distribution of health personnel between rural and urban areas. Traditional medical practitioners in Nigeria use a variety of herbal preparations to treat different kinds of ailments such as typhoid and paratyphoid fevers, dysentery, malaria, diarrhea, etc. Various research works conducted in recent years have shown that pathogenic microorganisms are becoming increasingly resistant to existing antibiotics at alarming rates (Rotimi and Mosadimi, 1984; Spencer et al., 1986; Olayemi and Oyagade, 1987; Monetiore et al., 1993, Olukoya et al., 1993). Such high wave of antibiotic resistance among pathogenic microorganisms creates problems in health care delivery in relation to microbial infections in man and animals. Piliostigma reticulatum (DL.) Hochst. (common name; Yoruba: ‘abafin’, Hausa: ‘kalgo’, Igbo: okpo atu’) belongs to the family Leguminosae - Caesalpiniaceae and is found in the savannah region of Nigeria. It is a tree, occurring up to 30ft in height with an evergreen, dense spreading crown (Keay, 1989). In recent past, attention has been directed towards medicinal research to substantiate the claims of cure made by traditional healers and thus provide scientific basis for their efficacy. Several important drugs have been discovered in plants and are now synthesized chemically for commercial purposes. However, the search for new ones continues especially in the tropical plants. Akinsinde and Olukoya, (1995) demonstrated activity of Terminalia avicennoides against Vibrio cholerae and Salmonella typhi, Omojasola and Awe (2004) reported the antimicrobial activity of the leaf extract of Anacardium occidentale and Gossypium hirsutum against Escherichia coli, Shigella dysenteriae, Salmonella typhimurium, Staphylococcus aureus and Pseudomonas aeruginosa. In Nigeria, the research for new drugs is on course hence the need to investigate the pharmacological potentials of Piliostgma reticulatum used traditionally in the treatment of diarrhea. Materials and Methods Sample Collection Sun dried bark of the plant was purchased from the trado-medical section of the Ilorin market. It was identified and authenticated at the Department of Plant Biology, University of Ilorin. Extraction Cold aqueous, hot aqueous and ethanolic extractions of the plant materials were prepared as described by Oyagade et al. (1999). A preliminary test had shown that the aqueous extract showed greater activity at 800C than 280C. So the aqueous extraction was done at 800C in water bath for 1½ hours. The extracts were decanted and filtered through Whatman No. 1 filter paper. The filtered extract was sterilized using 0.45µm pore size membrane filter and evaporated to dryness at 450C. The residues obtained were reconstituted in 95% ethanol as stock concentration of 250mg/ml. Test Organisms Clinical isolates of Escherichia coil, Shigella dysenteriae, Salmonella typhimurium, Staphylococcus aureus and Pseudomonas aeruginosa were collected from the University of Ilorin Teaching Hospital, Ilorin, Nigeria. All inocula were standardized using the method described by Bauer et al. (1966). Antibacterial assay The well method of the agar dilution was used to determine the antibacterial activity of the plant extracts and the antibiotics. The bacterial cultures were inoculated on Mueller Hinton Agar (MHA) and were incubated at 37OC for 24 hours. The MIC for the plant extracts was determined using the method of Mann et al., 1997 and Oyagade et al., 1999). Phytochemical screening of ethanolic extracts The extract was subjected to phytochemical screening for the presence of alkaloids, tannins, phenolics, saponins, steroids, triterpenes, phlobatinin and cardiac glycosides using the method described by Odebiyi and Sofowora, (1978). Results and Discussion The antibacterial activity of the cold aqueous, hot aqueous and ethanolic extracts of P. reticulatum show that the ethanolic extract exhibited greater activity than both the hot and cold aqueous extracts (Table 1). The higher activity of the ethanolic extracts may not be unconnected with the extraction solvent because ethanol has been shown to be better solvent than water (Akinyemi et al., 2000). This may also account for the reason why the herbal physicians prefer to use local alcoholic decoctions in extracting the herbs rather than aqueous extracts in treating their patients. The ethanolic extract was strongly positive for the presence of alkaloids, tannins and phenolics and positive for the presence of triterpenes and phlobatinins (Table 2). The presence of these phytochemicals is indicative of antibacterial activity (Odebiyi and Sofowora, 1978). The potential of any drug is the active principle contained therein; many of these secondary metabolites have prominent effect on animal systems and microbial cells (Jung et al., 1990; Triguna et al., 1992 and Fatoba et al., 2003). The presence of these metabolites therefore confers pharmacological potentials on this plant. Other workers have also reported bioinsecticidal and antitrypanocidal properties of P. reticulatum extract (Atawodi et al., 2003, Abdullahi and Muhammed, 2004). The cold water extract had least inhibitory effect on all the organisms used. The MIC ranged from 0.025 – 0.25 %w/v with P. aeruginosa having the highest value of 0.25%w/v (Figure .1). In the comparison of the antibacterial activity of P. reticulatum with some conventional antibiotics, the results indicate that the conventional antibiotics (Chloramphenicol, Ampicillin and Tetracycline) demonstrated greater activity than the plant extract at the same concentration (Fig. 2). The results also show that these differences are significant at 5% significance level. The fact that the plant extract is only a crude extract may account for some of these differences in activity. Higher levels of antibacterial activity are expected from a purified sample of P. reticulatum whose active principle has been isolated. The comparison of the activity of the plant extract with conventional antibiotics confirmed reports by other workers. Emeruwa (1982) reported that conventional antibiotics are more active than plant extracts. These results thus confirm the basis of traditional use of P. reticulatum in diarrhea treatment. Further study needs to be carried out on the isolation of the bioactive components of the tested plant and their effect in vivo. References Abdullahi, Y.M. and Muhammed, S. (2004). Assessment of the toxic potentials of some plants powders on survival and development of Callosobuchus maculatus. African Journal of Biotechnology 3(1):60 – 62. Akinside, K. A. and Olukoya, D. K. (1995). Vibriocidal activities of some local herbs. Journal of Diarrhoea Disease Research. 13: 127-129. Akinyemi, K. O., Coker, A. O.; Bayagbon C., Oyefolu, A.O.B., Akinside, K. A. andOmonigbehin, E. O. (2000). Antibacterial screening of five Nigerian medicinal plants against S. typhi and S. paratyphi. Journal of the Nigerian Infection ControlAssociation 3 (1). Atawodi, S.E., Bulus, T., Ibrahim, S., Ameh, D.A., Nok, A. J., Mamman, M. and Galadima, M. (2003). In vitro trypanocidal effect of methanolic extract of some Nigerian savannah plants. African Journal of Biotechnology. 2(9):317 – 321. Bauer, A. W., Kirby, W. N. Shervis, J. G. and Turrk, M. (1966). Antibiotics Susceptibilitytesting by a Standardized Single disc method. American Journal of Clinical Pathology: 45: 493. Emeruwa, A. L. (1982). Antimicrobial Substance from Carica papaya fruit extracts. Journal of Natural Product 45:123-127. Fatoba, P. O., Omojasola, P. F., Awe, S. and Ahmed, F. G. (2003). Phytochemical Screening of Some Selected Tropical African Mosses. NISEB Journal 3(2): 49-52. Jung, J. H., Pummangura, S., Onaichantipyuth, C., Patarapanich, C. and Mclanaghlin, J. L. (1990). Bioactive constituents of Melodrum fructicosum. Phytochem. 29(5): 1667-1670. Keay, R.W.J. (1989). Trees of Nigeria. Oxford Science Publication. pp 93, 194-196, 369. Montefiore, D.; Adeyemi-Duro, F.A.B. and Rotowa, N. A. (1993). Activity of Mezlocillin against In-patient strains of Pseudomonas aeruginosa and Proteus sp. West African Journal of Medicine 2(4):153-157. Oyagade, J.O., Awotoye, O. O., Adewumi, J. T and Thorpe, H.T (1999). Antimicrobial activity of some Nigeria medicinal plants. Bioscience Research Communications 11(3): 193 -197. Odebiyi, A. and Sofowora, A. E. (1978). Phytochemical Screening of Nigerian Medicinal Plants, Part III Lloydia 41:234-246. Olayemi, A. B. and Oyagade, J.O. (1987). Incidence of antibiotic resistance among E. Coli isolated from clinical source and river water Nigerian Medical Journal 17(4): 207-209. Olukoya, D. K., Idika, N. and Odugbemi T. (1993). Antibacterial activity of some medicinal plants from Nigeria. Journal of Ethnopharmacology 39: 69-72. Omojasola, P. F. and Awe, S. (2004). The antibacterial activity of the leaf extracts of Anacardium occidentale and Gossypium hirsutum against some selected microorganisms. Bioscience Research Communications. 60 (1): 25-28. Rotimi, V. O. and Mosadimi, H. A. (1984). Susceptibility of oral Bacteriods and Fusobacterium species to ten antibiotics. West African Journal of Medicine 3(2): 97-103. Spencer, E. E. E., Akuma, A. and Uduak, T. (1986). Surgical wound infection rate in Calabar University Teaching Hospital. West African Journal of Medicine 5(1): 61-68. Triguna, N. M., Singh, R. S., Pandey, H. S., Prasad, C. and Singh, B. P. (1992). Antifungal essential oil and a long chain alcohol from Achyranthes aspera. Phytochem. 31(5): 1811 – 1812. WHO (1978). The Promotion and development of traditional medicine. World Health Organization, Technical Report Series No. 622.
Table 1. Antimicrobial activity of the bark extract of Piliostgma reticulatum.
Table 2. Phytochemical Screening of bark extract of P. reticulatum.
Key: ++ Strongly positive, + Positive, - Not detected.
Figure 1. Minimum Inhibitory Concentration (MIC) of Ethanolic extract.
Key: P.RET - P. reticulatum, CHLO-Chloramphenicol, AMP- Ampicillin, TETRA- Tetracycline. Fig .2 Antibacterial activity of ethanolic extract compared with the conventional antibiotics.
|
||||||||||||||||||||||||||||||||||||||||||