|
Ethnobotanical Leaflets 12: 476-480. 2008.
Inter Species Relationship of Cinnamomum Species Using RAPD Marker Analysis
Priya joy1 and M. Maridass2
1Centre for Biodiversity and Biotechnology, 2Animal Health Research Unit St. Xavier’s College (Autonomous), Palayamkottai-627002, Tamil Nadu, India Corresponding author: E-mail [email protected] Issued 13 July 2008
Abstract Genetic inter-relationship of nine Cinnamomum species was estimated using randomly amplified polymorphic DNA (RAPD) markers. The 15 selected RAPD primers out of 2 primers were amplified in all cinnamon species. Cinnamomum verum has very close similarity (87%) with C. citronella and another two species of C. camphora and C. glucens. Our results suggested that genetic relationships in Cinnamomum species using RAPD banding data may be useful for plant improvement and an efficient way to conserve genetic resources of Cinnamomum species, in addition to their effective medicinal uses. Key Words: Lauraceae; Cinnamomum; DNA; RAPD; Southern India. Introduction This manuscript examines the standardization of RAPD marker and genetic relationships of Cinnamomum species, a commercially valuable source of camphor, cinnamaldehyde and safrole oil in the world. Cinnamomum the largest genus in the Lauraceae, it has comprises 250 species, which are distributed in India,SriLanka and Australia. Cinnamomum was first introduced in India from Sri Lanka. Nineteen species of Cinnamomum have been found on the South India. Many species of Cinnamon yield a volatile oil on distillation. The most important Cinnamon oils in world trade are those from C. zeylanicum, C. cassia and C. camphora. The other species given oils, which are utilized as sources for chemical isolates. However, a number of other cinnamomum species are distilled on a much smaller scale and the oils used both locally or exported (The Wealth of India, 1992).The major compounds present in both stem-bark and root bark are cinnamaldehyde (75%) and camphor (56%), respectively (Senanayake et al., 1978). Cinnamon bark oil possesses the aroma of the spice and a sweet and pungent taste. It is employed mainly in the flavorings industry where it is used in meat and fast food seasonings, sauces, pickles, baked goods, confectionery, cola-type drinks, tobacco flavors and dental and pharmaceutical preparations. Many species of Cinnamomum are known to contain safrole (Bin Jantan and Goh, 1992), and concern that this beverage contains a known carcinogen led to this study. Recently global interest in oriental medicine, production of those plants has grown even more over the following years. Since many species and varieties exist, development of molecular markers would be important for quality assessment in the medicinal industry (Sang-Bok Lee, et al, 2000). During the last decade several novel DNA-markers (RAPD, RFLP, SSR, ISSR etc.) have been rapidly integrated into the tools available for genome analysis. Salimath et al., (1995) has been used for DNA fingerprinting and assessing genetic diversity. Presence or absence of DNA bands in the gel may be used as RAPD markers to study close genetic relationship (Sang-Bok Lee, et al., 2000), inter- and intra-specific genetic variations (M’Ribu and Hilu 1994), for the identification of specific genes (Paran et al. 1991; Martin et al. 1991), and to study the pattern of gene expression (Valle et al. 2000). Materials and Methods Genetic material Young leaves of nine Cinnamomum species viz.,C.camphora C. cassia, C. riparium, C. macrocarpum, C. perrotteiti, C. wightii C. citronella, C. glaucense, C. verum C.malabaratum were collected from Southern India and preserved at – 80°C.
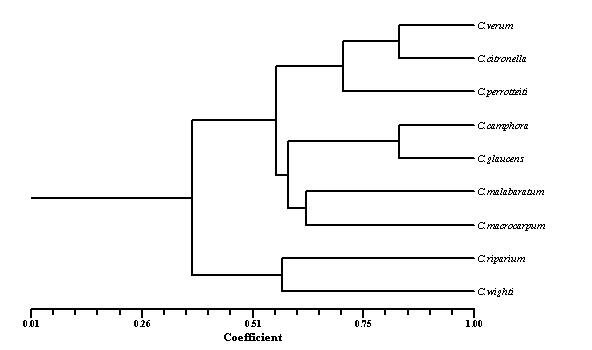
DNA extraction Stored leaves were pulverized in liquid nitrogen and DNA was extracted from each plant of Cinnamomum species according to the method described by Doyle and Doyle (1987). Total DNA was quantified spectrophotometrically and samples yielding good quality (A260/A280 ratio 1.7–1.9) and as well as visually by ethidium bromide staining on 0.8% agarose gel electrophoresis. RAPD Marker analysis A set of random 15 primers was purchased from a commercial source (Operon Technologies, Alameda, Calif.). After initial tests, 15 primers (OPA-10; OPA-12; OPA-15; OPA-17; OPB-01; OPB-15; OPC-01; OPC-15; OPE-02; OPE-06; OPE-07; OPE-10; OPE-20; OPF-13; and OPF-20) were used for further studies. PCR reactions were performed according to the protocol of Williams et al. (1990). Briefly, DNA samples of the 10 Cinnamomum species of each individuals plants were adjusted to 20 ng/µl and used in the amplification reactions with a final volume of 25µl containing 1µl of DNA, 2 µl of primer (40 µM), 1 µl of dNTPs (10 mM), 0.2 µl Taq DNA polymerase (5 U/µl), 3 µl PCR buffer, 1.5 µl of MgCl2 (25 mM) and 16.3 µl dionized water. DNA amplification was carried out using a eppendrof thermocycler programmed with 3 min at 94°C for initial denaturation, followed by 35 cycles of 54s at 94°C, 45 sec at 43°C, 2 min at 72°C, and a final 5 min extension at 72°C. After amplification, the DNA fragments were separated by electrophoresis for about 2 h under constant voltage (60 V) in 1.8% agarose gel submersed in 1X TBE buffer. All PCR experiments were done at least twice and the best gels of the replicates were used for band scoring. The gels were stained with ethidium bromide solution and observed under ultraviolet light. Each gel was photo documented using the image capturing system bioprint. A 1 kb fragment size marker was used as a reference to allow comparison among the different gels (Gibco 1kb ladder). Statistical analysis The amplified bands were scored as 1 and 0 based on band (allele) presence and absence, respectively. Sizes of amplified bands were estimated using Gel Pro analyzer software. The binary data set was used to calculate the pairwise Jaccard similarity index and to assemble the corresponding similarity matrix. The matrix obtained was used to generate a dendrogram using the UPGMA method (Unweigthed Pair Group Method Arithmetical Means). The distances in the dendrogram were compared with the genetic distances between genotype pairs to calculate the cophenetic correlation. All the analyses were performed with the aid of the 1.70 version of the NTSYS-pc computer program. Results and Discussion Fifteen primers were used in this study of RAPD marker analysis to standardization of suitable specific primers amplifying the genetic materials of Cinnamomum species. All primers but two (OPE-20, and OPE-13) yielded maximum amplification products with all Cinnamomum species. Primers OPE-20 and OPE-13 amplifying all Cinnamomum species were produced many bands overall ranging in size 600 - 2600bp. The primers amplified DNA products from each Cinnamomum species generating reproducible band patterns. Primers OPE-20, and OPF- 13 generated uncommon band with DNA from some of the specimens, along with several common bands with each specimen .Primer OPE-20 was amplified in C.verum very close similariety (87%) with C.citronella, C.camphora and C.glucens. Several polymorphic bands were detected. The remaining primers gave patterns that were identical or had differences too small to provide information on the genetic diversity. They could hardly distinguish the nine Cinnamomum species slightly different all Cinnamomum species. A phylogenetic tree comprising a total of 9 Cinnamomum species RAPD marker was constructed as shown in Figure 1. The correlation coefficient calculated between RAPD when using the similarity 87% when using the dendrograms.
Fig. 1: Dendrogram showing OPA - 20 primers using analysis of inter species relationship of Cinnamomum species
Cinnamomum species is a valuable spices tree in India, Sri Lanka Asia, and Australia. Driven by commercial incentives, the wild population of this tree has been threatened with depletion in recent years due to excessive harvesting. The present study was preliminary attempt to develop RAPD primers to distinguish the nine Cinnamomum species showed that a more difficult screening of primers has to be done before RAPD markers can be developed. Chatti et al. (2003) work done on the genetic diversity and the phylogenetic relation between 17 ecotypes analyzed by using the technique of RAPD marker. This study showed a significant morphological variation and a large genetic diversity within and among cultivars. Lately, this technique has been used to study the genetic relations between the different species of coffee and to determine the relationship between hybrids (Paulo, 2003). The dendrogram supports the suggestion that similar variation of C. verum and C. citronela and also may be synonymous with other two species of C. riparium and C. wighti . However the variation in terms of length 600-2600 bp and nucleotide the frequency of insertions and deletions is too large for phylogenetic analyses. Acknowledgments We ould like to thank the State Forest Department, Tamil Nadu and Kerala for permission to collect plant materials.
References Bin Jantan, I., Goh, S.I., 1992. Essential oils of Cinnamomum species from peninsular Malaysia. Journal of Essential Oil Research 4, 161–171. Doyle and Doyle (1987). A rapid DNA isolation procedure for small quantities of fresh leaf tissues. Phytoch. Bull. 19: 11-15. M’Ribu, H.K., and Hilu, K.W. 1994. Detection of interspecific and intraspecific variation in Panicum millets through random amplified polymorhic DNA. Theor. Appl. Genet. 88: 412–416. Paran, I., Kesseli, R., and Michelmore, R. 1991. Identification of RFLP and RAPD markers linked to downey mildew resistance gene in lettuce using near-isogenic lines. Genome, 34: 1021–1027. Salimah, S.S., De Oliveira, A.C., Godwin I. D (1995). Assessment of genomic origin and genetic diversity in the genus Eleusine with DNA markers. Genomic. 38: 757–763. Sang-Bok Lee, and Søren K. Rasmussen, 2000. Molecular markers in some medicinal plants of the Apiaceae family. Euphytica, 114: 87–91. Senanayake U. M., Lee T. H. and Wills R. B. H. (1978), Volatile constituents of cinnamon (Cinnamomum zeylanicum) oils. J. Agric. Food Chem. 26, 822-824. The Wealth of India, (1992). A Dictionary of Indian Raw materials and Industrial products, III Publications and Information Directorate, New Delhi, p. 582-590. Valle, P.R., Souza, M.B., Pires, E.M., Silva, E.F., and Gomes, M.A. 2000. Arbitrarily primed PCR fingerprinting of RNA and DNA in Entamoeba histolytica. Rev. Inst. Med. Trop. Sao Paulo, 42(5): 249–253. Williams JGK, Kubelik AR, Livak KJ, Rafalski JA and Tingey SV (1990) DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Research 18:6531-6535.
|