|
Ethnobotanical Leaflets 12: 96-102. 2008.
The Petiolar Structure of Christella dentata (Forssk.) Brownsey & Jermy (Thelypteridaceae, Pteridophyta)
KAMINI SRIVASTAVA, M.Sc, D.Phil Department of Botany, University of Allahabad, Allahabad-211002 India E-mail: [email protected] Issued 25 February 2008
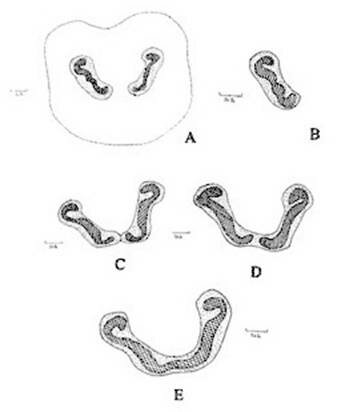
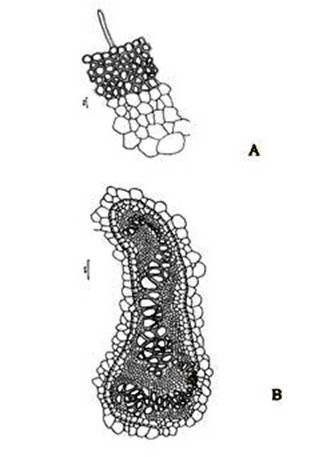
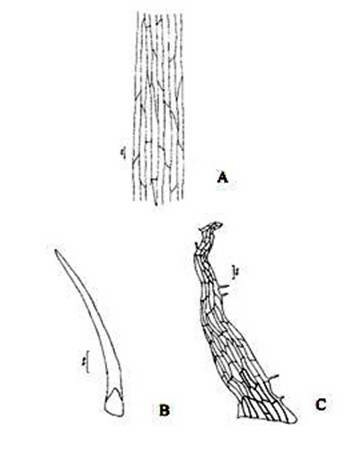
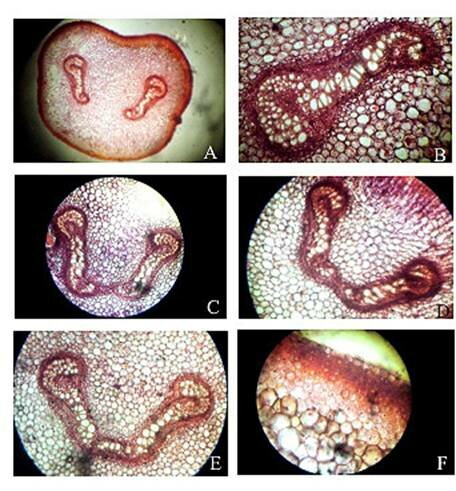
Abstract The exomorphic and structural details of the petiole of Christella dentata, a fern frequently grown in gardens and glass houses as an ornamental, are illustrated here for the first time. The stipes receive two separate vascular strands from the rhizome which fuse together to form a single strand during their upward course. Key words: Christella dentata, petiole anatomy, vascular strand. Introduction Fern taxonomists have long employed the characteristics of the rhizome, indusium, sporangia and spores in their construction of the systematic treatments of various fern taxa. Leaf characters like venation pattern, trichomes, epidermis and ontogeny of stomata have also been utilized for taxonomic purposes. Likewise, stipe characters have also proved to be of great value (Bower 1914, 1926; Kato 1972; Ogura 1972; Lucansky and White 1974; and Linn and Devol 1977, 1978). However, the characteristics of the stipe still remain to be studied in the case of a number of fern species, and especially of the Indian forms. With this thought held firmly in mind, the author has chosen to study the exomorphic and internal details of stipe of Christella dentata. Similar investigations for other species of Christella viz.C. cylindrothrix, C. subpubescens and C. malabariensis have been made by Shankar (1986). Materials and methods Plants of C. dentata were obtained from the fern house of the Botany Department of Allahabad University, India. Pieces of stipe were fixed in F.A.A and sectioned in different planes either by hand or with the help of a microtome set at 10-12 mm thickness. Sections were stained with safranin or safranin fast green combinations. Transparencies were also made by clearing the petioles in 10% aqueous sodium hydroxide solution and chloral hydrate and subsequently staining with safranin (Foster 1966). To confirm the nature of various depositions and cell substances, special micro and histochemical tests were performed, e.g., lignin and cutin were tested by obtaining red colour with pholroglucinol and sudan IV and starch by blue colour with iodine solution respectively (Johansen 1940). For phlobaphenes, a histochemical test was performed as suggested by Reeve(1951). Observations The petiole of C.dentata is cylindrical. On the adaxial side of the petiole there is a prominent groove running from the base up to the tip where the first pinna is attached. The base of petiole is usually covered by scales. The marginal cells of scales are provided with fine projections (Fig. 3 C). In addition to simple scales, elongated hairs with tapering ends are found (Fig. 3 B). In surface view of the petiole, epidermal cells appear narrow and elongated and devoid of stomata (Fig. 3 A). In a transverse section of the petiole, epidermal cells throughout their length appear small, thick walled, dark brown in colour and covered by smooth and delicate hairs. The epidermis is followed by three or more layers of thick walled cells followed by several layers of thin walled parenchymatous cells which constitute the ground tissue (Fig. 2A; Plate 1 F). Microchemical tests reveal that the entire ground tissue is made up of cellulosic cells. Thin walled inner layers of cells usually have starch grains. The petiole receives two widely separated vascular strands from the rhizome (Fig. 1A; Plate 1A). Each vascular strand is enclosed by a single layered endodermis. The pericycle is made up of thin walled cells, which are one to three layers in thickness. The xylem is mesarch and surrounded by phloem. The xylem is hippocampus-shaped and the two vascular strands appear slightly elongated. The adaxial arms are comparatively more turned inwards and the two vascular stands appear almost reniform in shape (Fig. 1A; Plate 1A). Soon after entering the stipe base, the two separate vascular strands start coming closer to each other and get fused somewhere near the middle of the petiole to form a single strand for further upward course(Fig. 1C; Plate 1C). During the merger of the two vascular strands first the endodermis, the pericycle and at still higher levels the two phloem and xylem strands also join each other at their abaxial side (Fig.1D, E; Plate1 D, E). The distal arms of xylem and phloem however remain as such. Thus, the single vascular strand that resulted due to fusion of the two is almost ‘U’ shaped and free arms of xylem strands turned inwards more in the former as compared to the latter (Fig. 1E; Plate 1E). Xylem consists of tracheids with protoxylem having annular or spiral thickenings and metaxylem with scalariform thickenings. Phloem consists of sieve cells with occasional parenchyma (Fig. 2B; Plate 1B). Conclusion and Discussion The petioles of C. dentata show differences in their external as well as internal structure as compare to C. cylindrothrix, C. subpubescens and C. malabariensis. Their bases are covered with scales consisting of thin walled cells. The marginal cells of scales have blunt projections in C. cylindrothrix and C. malabariensis and pointed in C. subpubescens and C. dentata. Multicellular hairs are distributed through out the petiole in all the three species except in C. dentata. From the base of the petiole a very shallow groove arises on their adaxial sides which gradually become obscure in the apical portion of C. cylindrothrix. However, in C. subpubescens, C. malabariensis and C. dentata it remains prominent even in the apical region of the petiole. Stomata are absent on the epidermal portion of petiole of C. dentata, C. cylindrothrix and C. subpubescens except in C.malabariensis. Petioles of all the four species receive two widely separated vascular strands from rhizome. Each vascular strand is enclosed by an outer endodermal layer whose cells lack the characteristic casparian thickenings and two to four layers of pericycle made up of thin walled cells. The xylem is measarch and surrounded by phloem. At certain places xylem was found intermixed with parenchyma. Phloem consists of sieve cells and parenchyma filled with tannin. Tannin cells are absent in C. dentata .The shape of vascular strand is slightly elongated with hippocampus shaped xylem in C. dentate, C. subpubescens and C. malabariensis and almost ‘C’ shaped in C. cylidrothrix with hippocampus shaped xylem strand. The proximal and distal arms of xylem in C. cylindrothrix is straight while it is turned inwards in C. dentata, C. malabariensis and C. subpubescens. The vascular strands in C. cylindrothrix and C. malabariensis remain as such throughout their course in the petiole while in C. dentata and C. subpubescens the two widely separated vascular strands, somewhere in the middle of the petiole join each other to form a single ‘U’shaped vascular strand with free arms of xylem turned inwards. During their fusion, first the adjacent sides of endodermis join each other followed by pericycle. Ultimately the xylem and phloem strands also join through the metaxylem of abaxial ends. This single vascular strand thus formed remains as such throughout the remaining course in the petiole.
Figure 1. Christella dentata. A. Diagrammatic transection of petiole at the base showing two vascular strands; B. One of the vascular strands in a magnified; C, D, E. Diagrammatic transection of petiole at various levels showing stages during fusion of the two strands.
Figure 2. Christella dentata. A. A portion of epidermis and cortex of petiole in a transaction; B. Structural details of a vascular strand.
Figure 3. Christella dentata. A. Surface view of epidermis; B. Hair from the petiole surface; C. Scale from the petiole surface.
Plate 1. Christella dentata. A. Transection of petiole at base showing two vascular strands; B. One of the two vascular strands in a magnified; C, D, E. Transection of petiole at various levels showing stages during fusion of the two strands; F. A portion of epidermis and cortex of petiole in a transaction.
References
BOWER, F. O.1914. Studies in the phylogeny of the filicales.1v. Blechnum and allied genera. Ann Bot. 28:363-431.
BOWER, F. O.1926. The Ferns. vol.II. London.
KATO, M. 1972. The vascular structure and its taxonomic significance in the Athyriaceae. Acta Phytotax. Geobot. 25: 79-91.
OGURA, Y.O. 1972. Comparative Anatomy of the Vegetative Organs of the Pteridophytes, Berlin, Stuttgart.
LUCANSKY, T.W. & WHITE, R.A. 1974. Comparative studies of the nodal and vascular anatomy in the neotropical Cyatheaceae, 3. Nodal and petiole patterns; Summary and conclusions. Amer. J. Bot. 61: 818-828.
LIN, B.L. & DEVOL, C.E. 1977. The use of stipe characters in fern taxonomy I. Taiwania. 22: 91-99.
LIN, B.L. & DEVOL, C.E. 1978. The use of stipe characters in fern taxonomy II Taiwania.23:77-95.
SHANKER, R.1986. Morphotaxonomic studies of some Pteridophytes. D.Phil.Thesis, Departement of Botany, TheUniversity of Allahabad, Allahabad.
FOSTER, A.S. 1966.Morphology of anstoses in the dichotomous venation of cercaeaster. Amer. J. Bot. 53: 588-599.
JOHANSEN, D.A. 1940. Plant Microtechnique. Mc Graw Hill Co. New York.
REEVE, R.M. 1951. Histochemical test for polyphenols in plants. Stain Technol. 26: 91-96.
|