|
Ethnobotanical Leaflets 13: 1485-1503, 2009. Studies on Phytochemical constituents, Functional Group Identification and Antimicrobial Activity of Solanum nigrum (Solanaceae)
*D.venkatesan1; C.M. Karrunakaran2; S. Selva kumar3
*1Department of Industrial Biotechnology, Bharath University, Chennai Tamil Nadu, India. Pin-600 073. Phone 91- 09003543949 2Dean-Research, Bharath University, Chennai Tamil Nadu, India. Pin-600 073, Phone 91-09443256172. E mail:[email protected] 3Lecturer, Bharath University, Chennai Tamil Nadu, India. Pin-600 073, Phone 91-09790956401. E mail: [email protected]
*Corresponding author:
Issued 01 December, 2009
Abstract
Phytochemical constituents, functional group identification and antimicrobial activity of ethanolic extract of Solanum nigrum were investigated. The phytochemical screening of the crude extract revealed the presence of alkaloids, reducing sugars, tannis, flavonoids, Phlobatannis, and steriods. The extract were subjected for identification of functional groups using infra red spectrophotometer, presence of C=O, C-H, C=C and C-O were identified, these bonding structures are responsible for presence of alkyl groups, methyl groups, alcohols, ethers, esters, carboxylic acid and anhydrides. And the crude ethanolic extract of Solanum nigrum were assayed for in vitro antimicrobial activity against gram positive and gram negative bacteria and they were also subjected for antifungal activity and the zone of inhibition were compared with control drug Penicillin and Nystatin. Staphylococcus aureus as the most sensitive strains exhibited maximum zone of inhibition about 25.7mm among gram positive organism tested. Escherichia coli exhibits 30.1mm zone than that of standard penicillin positive control. Trichophyton mentagrophytes exhibits maximum zone of inhibition of about 26.1mm among the tested fungal species. Key words Solanum nigrum, Staphylococcus aureus, Escherichia coli, Trichophyton mentagrophytes, Infra red spectrophotometer. Introduction Natural products drug discovery will be more holistic, personalized and involve wise use of ancient and modern therapeutic skills in a complementary manner so that maximum benefits can be occurred to the patients and community (Patwardhan and Hopper, 1992). The antiseptic qualities of aromatic and medicinal plants and their extracts were recognized in the laboratory data back to the early (Dorman and Dean, 2000). As result of ecological and fitgeographical diversification, endemism of species is high (Tan, 1992; Dagic et al., 2002). Ayurvedic remaining one of the most ancient and yet living traditions practiced widely in developing countries as well as developed countries and has a sound philosophical and experimental basis (Dahanukar and Thathe, 2000; Chopra and Doiphode, 2002). In recent years investigation found several plants of the ethnomedicine posses really interesting biological activities which could be of interest for all parts of the world (El- Fiky et al., 1995; Awadh Ali et al., 2001; Mothana and Lindequist, 2005; Mothana et al., 2006; Al-Fatimi et al., 2007). Approximately 20% of the plants found in the world have submitted to pharmacological activities (Suffrendini et al., 2004). And the plants have organic chemicals (Evans et al., 1986). The plants posses chemotherapeutic, bacteriostatic and antimicrobial agents (Purohit and Mathur, 1999) The source of plants is models for the synthesis of new drugs with better therapeutic, chemical (or) physical properties than the original compound (Baerheim svendsen and Scheffer, 1992). In recent years plant material have been investigated as a source of medicinal agents (Krishnaraju et al., 2005). And the plants materials used for treatment bacterial infections (Balandrin et al., 1985). However world health organization also has recognized the importance of traditional medicine and has been active in crating strategies, guidelines and standards for botanical medicines (WHO, 2002). Plant derived drugs remains important resource especially in developing countries, to combat serious disease. Approximately 62 80% of the worlds population still relies on traditional medicines for the treatment of common illness (Zhang, 2004). In fact, plants produce a diverse range of bioactive molecules making them a rich source of different types of medicines. Higher plants, as sources of medicinal compounds, have continued to play a dominant role in the maintenance of human health since ancient times (Farombi, 2003). Over 50% of all modern clinical drugs are of natural product origin (Stuffness and Douros, 1982). And natural products play on important role in drug development programmes in the pharmaceutical industry (Baker et al., 1995). There are a few reports on the use of plants in traditional healing by either tribal people or indigenous community (Sandhy et al., 2006; Ayyanar and Ignacimuthu, 2005; Rajan et al., 2002; Natarajan et al., 1999; Ignacimuthu et al., 1998). The activities have been have been selected because of their great medicinal revelence with the recent years, infections have increased to a great extent and resistant against antibiotics, becomes an ever increasing therapeutic problem (Austin et al., 1999). Because natural products of higher plants may give a new source of antimicrobial agents. There may research group that are now engaged in medicinal plants research (Samy et al., 1998; Hamil et al., 2003; Motsei et al., 2003). The development drug resistance in human pathogens against commonly used antibiotics has necessitate a search for new antimicrobial substance from other sources including plant screening of medicinal plants for antimicrobial activities and photochemical is important for finding potential new compounds for therapeutic uses. In present study focus phytochemical constituent, functional group identification and in vitro evaluation of antimicrobial activity of the separated fractions of ethanolic extracts from the aerial parts of Solanum nigrum are bioassay against pathogenic microrganism. Materials and Methods Collection of Plant materials and Identification Plant material of Solanum nigrum was purchased from a local Ayurvedic medicinal shop "Chennai," and the material was identified and authenticated by the Chief Botanist, Tamil Nadu Aromatic Medicinal Plants Corporation Limited (TAMPCOL), Arignar Anna Siddha Medical College and Hospital Campus, Chennai, Tamil Nadu, India. Ethanolic extraction The plant materials were dried in shaded and powdered by mechanical grinder. The powder of Solanum nigrum plant material were initially defated with petroleum benzene ( 60 - 80C) followed by 1000 ml of ethanol by using a Soxhlet extractor for 72 hours at a temperature not exceeding the boiling point of the solvent. The extract was filtered using whattman filter paper (No 1) and then concentrated in vaccum and dried at 45C for ethanol elimination and the extract were kept in a sterile bottle under refrigeration condition of about 2-8C. Test for Phytochemical analysis The extracts were analyzed for the presence of alkaloids, terpenoids, reducing sugars, Saponins, tannis, Carbonyls, Flavonoids, Phlobatannis and steriods (Adetuyi and Popoola 2001; Trease and Evans, 1989; Sofowora, 1982). Test for Alkaloids Weigh about 0.2 gm of plant extract in separate test tube and warmed with 2% Sulphuric acid for 2 minutes. And it was filtered in separate test tube and few drops of Dragencloffs reagent were added and observed for the presence of orange red precipitates for the presence of alkaloids. Test for Cardiac glycoside Keller-Killani Test Weigh about 0.5 gm of plant extract in a separate test tube with 2 ml of glacial acetic acid containing a drop of ferric chloride solution. This was under layered with 1 ml of concentrated tetra oxo sulphate (VI) acid. And observe for brown ring formation at the interface (Finar, 1983). Test for Terpenoids Weigh about 0.5 g plant extract in separate test tubes with 2 ml of chloroform. And add concentrated Sulphuric acid carefully to form a layer. And observe for presence of reddish brown color interface was formed to show positive results for the presence of terpenoids. Test for reducing sugars Take a test tube and add 2 ml of crude plant extract and add 5 ml of Distill water and filtered. The filtrate was boiled with 3-4 drops of Fehlings solution A and B for 2 minutes. Observe for orange red precipitate indicates the presence of reducing sugars. Test for Saponins Weigh about 0.2 gm of plant extract in the test tube and add 5 ml of distill water and then heat to boil. Observe for frothing (appearance of creamy mass of small bubbles) shows the presence of Saponin Test for Tannis To small quantity of plant extract was mixed with water and heated on water bath. The mixture was filtered and ferric chloride was added to the filtrate. And observe for dark green solutions indicates the presence of tannis Test for Carbonyl Take 2 ml of plant extract in separate test tubes and add few drops 2,4, di nitro phenyl hydrazine solution and shaken. And observe for the presence of yellow crystals immediately for the presence of an aldehyde. Test for Flavonoids Weigh about 0.2 gm plant extract in separate test tubes and dissolved diluted Sodium hydroxide and add diluted Hydrochloride. And observe for yellow solutions that turns colorless indicates the presence of flavonoids Test for Phlobatanin Weigh about 0.5 gm of plant extract in a test tubes and dissolved with distill water and filtered. The filtrate was boiled with 2% Hydrochloric acid solution. Observe for red precipitate shows the presence of Phlobatanin Test for Steroids To the plant extract add 2 ml of acetic anhydride and add 0.5 gm of ethanolic extract of each sample with 2 ml of Sulphuric acid .Observe for the color change from violet to blue or green in samples indicating the presence of steriods Identification of functional group Instrument used : FTIR (Fourier Transformer Infra red Spectrophotometer) Make : Shimadzu Sample preparation for Infrared Spectrophotometer A drop of ethanolic extract of Solanum nigrum was placed on the Sodium chloride cell to obtain the thin layer. Then the cell was placed in the FTIR compartment and scanned accordance with ASTM (American standard testing material) 1252-98 through IR region for about 25 scans and chromatogram was obtained. Antibacterial activity Bacterial strains and Growth conditions The following cultures of gram positive organism were used Streptococcus pyogenes, Streptococcus agalactiae, Streptococcus faecalis, Staphylococcus aureus, Staphylococcus epidermidis, Bacillus anthracis, Bacillus subtilis and gram negative organism of Escherichia coli, Klebsiella pneumoniae, Shigella flexneri, Shigella sonnei, Shigella boydii, Salmonella typhi, Salmonella paratyphi, Vibro cholera and Pseudomonas aeruginosa. The fungal cultures used were Microsprum gypsum, Trichophyton mentagrophytes, Epidermophyton floccosum and Aspergillus niger. The cultures are obtained from National Collection of Industrial Microorganism (NCIM) Pune, India. The bacterial cultures were maintained in the nutrient agar slants and fungal cultures were maintained in Sabouroud dextrose agar slant at 2 - 8C. Preparation of plant extract, antibiotic and Inoculum preparation The dried plant extract were weighed and dissolved in sterile distilled water to prepare appropriate dilution to get required concentration of 5.0 mg/ml and antibiotic at 50g/disc. Inoculum of bacterial cultures were prepared in nutrient broth medium and kept incubation at 35C for 8 hours and fungal cultures at 25C for 24 hours. Procedure for performing the Disc Diffusion test (Bayer et al., 1986) The required amount of Mueller-Hinton plates (Hi media) is prepared as per manufacturer instructions. A sterile cotton swab was dipped into the turbid culture suspension. The dried surface of Muller-Hinton agar plate was inoculated by streaking two more times rotating the plate approximately 60each time. The lid may be left aside for 3-5 minutes and allow to dry for the excess surface moisture content. The ready prepared sterile Himedia discs (0.7mm) were poured with different concentration of above prepared antibiotic and plant extract solutions, the discs were placed on the medium and the plate were incubated at 5C for 1 hour to permit good diffusion and then transferred to an incubator at 37C for 24 hours for bacterial cultures and for fungal cultures the plates should be incubated at 25C for 72 hours. The negative control was included without adding the cultures to know the sterile conditions. The antibacterial activity was recorded by measuring the width of the clear inhibition zone around the disc. Results and Discussion Ayurveda remains one of the most ancient and yet living traditions practiced widely in India. Medicinal herbs consist of wide variety of chemical compounds nearly 80% of the world population depends upon traditional system of health care. Survey had revealed that 50% of the top prescription drugs in the world are based on natural products. Hence this present study was conducted to study the in vitro antimicrobial activity of medicinal plant used by Indian peoples to show that the therapeutic properties of Solanum nigrum used in traditional medicine coincide with laboratory findings. The ethanolic extract of Solanum nigrum were subjected for phytochemical analysis, the results shows that positive for tests of alkaloids, reducing sugars, tannis, flavonoids, phlobatannis, and steriods but shows negative results for cardiac glycosides, terpenoids, saponins and carbonyl were discussed in the table -1 Table 1. Phytochemical constituents of Solanum nigrum.
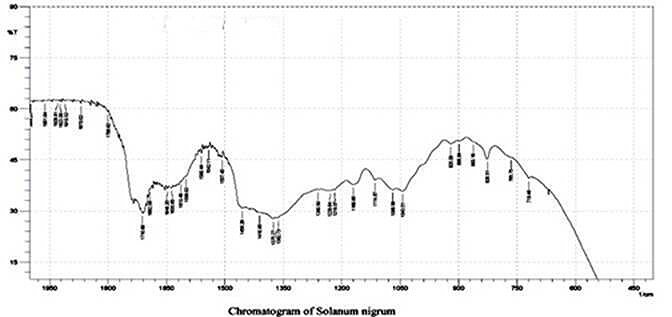
Key + = Positive ; - = Negative The Infra red spectroscopic (IR) analysis of ethanolic crude extract of Solanum nigrum, reveals that the presence of different functional group ranging from 4000 to 400 wave number, C-Hstrl, alkyl group, (2926.6cm-1), C=O stretching for carbonyls (2162.1 cm-1), C-O bending for alcohols, ethers, esters, carboxylic acid and anhydrides (1310.6 1059.6cm-1), C-H bending alkyl (1453.4 1376.2 cm-1), C=C stretching (1660.0- 1667.0) and C-H bending for methyl group (864.1 668.4 cm-1) The spectroscopic analysis of the plant extract of Solanum nigrum, figure -1 shows peaks range from 2000 400 wave number were discussed in the table -2 the extract revealed the presence of C=O, C-H, C=C and C-O bond stretching. Generally O-H stretching will be present in all phenolic compounds and C-N is commonly found in all types of Alkaloids. Table 2. IR Spectroscopic analysis of ethanolic extract of Solanum nigrum, which showing characteristic absorption peaks at IR range.
Figure 1. IR Spectroscopic analysis of ethanolic extract of Solanum nigrum.
The in vitro antimicrobial activity of ethanolic extract of Solanum nigrum was assessed qualitatively and quantitatively by the presence or absence of inhibition zones, Zone diameter values, the ethanolic extract of Solanum nigrum showed great in vitro antimicrobial activities against 16 types of bacteria and 4 fungus species tested. In this study the antimicrobial activities of ethanolic extract were compared with standard antibiotics such as Penicillin and Nystatin used as positive control. Results from the antimicrobial disc diffusion assay were furnished in table-3, the data indicates that gram positive Staphylococcus aureus was the most sensitive strain tested to the ethanolic extract of Solanum nigrum with greatest inhibition zone of about 25.7mm. Staphylococcus epidermidis, Bacillus anthracis and Bacillus subtilis were also found more sensitive with zone of inhibition 22.3mm, 20.1mm and 22.1mm. Streptococcus pyogenes show less activity when compared with control standard standard penicillin with zone of inhibition about 10.2mm. The ethanolic extract of Solanum nigrum also shows excellent activity against Escherichia coli, tested gram negative organism with 30.1mm zone of inhibition to that of standard penicillin positive control. Klebsiella pneumoniae, Shigella flexneri, Salmonella typhi and Pseudomonas aeruginosa exhibited maximum significant zones to the tested ethanolic crude extract of Solanum nigrum with inhibited zones of about 28.1mm, 26.0mm, 18.2mm, and 21.5mm. Shigella boydii, Salmonella paratyphi and Vibro cholera exhibited better significant zones of about 10.0mm, 09.1mm and 12.5mm. The ethanolic extract of Solanum nigrum also exhibited the highest antifungal activity against Microsporum gypsum, Trichophyton mentagrophytes, Epidermophyton floccosum and Aspergillus niger with the inhibition zone of 24.2mm, 26.1mm, 22.1mm and 18.7mm when compared with control standard Nystatin Table 3. Antimicrobial activity expressed as minimum inhibitory concentration (MIC) of ethanolic extract of Solanum nigrum by disc diffusion method.
Note Values in the means of triplicate Ethanolic extract concentration 5.0 mg/ml Penicillin 50g/disc Nystatin 50g/disc Conclusion This research work states that the presence of presence of alkaloids, reducing sugars, tannis, flavonoids, Phlobatannis, and steriods are responsible for the antimicrobial activity, the ethanolic extract of Solanum nigrum have of C=O, C-H, C=C and C-O bond stretching compounds exhibits maximum zone of inhibition against the tested organism when compared with control drug penicillin and Nystatin .Hence present study suggests that pathogenic microorganism become resistant to the existing drugs. And this study shows that the plant is promising development of phytomedicine for antimicrobial properties. In this endeavor, traditional herbal medicines must perforce be granted the benefits of modern science and technology to serves further global needs. The drugs derived from herbs may have the possibility of using in medicine because of its antibacterial activity. Acknowledgements We wish to acknowledge and thankful to Dr. C.M. Karrunakaran, Ph D., Dean-Research, Bharath University for their valuable and consistent support and guidance for completion of this work. Reference
Adetuyi, A.O and Popoola, A.V. (2001). Extraction and dyes ability potential studies of the colourant in zanthoxylum zanthoxyloides plant on cotton fabric, Journal of Science Engineering Technology 8 (2): 3291-3299. Al-fatimi, M., Wuster, M., Schroender, G., Lindequist, U. (2007). Antioxidant antimicrobial and cytotoxic activities of selected medicinal plants from Yemen. J.Ethnopharmacol 111: 657-66. Austin, D.J., Kristinsson, K.G., Anderson, R.M. (1999). The relationship between the volume of antimicrobial consumption in human communities and the frequency of resistance. Proc Natl Acad Sci USA 96:1152-6. Awadh Ali, N.A., Juelich, W-D., Kusnick, C., Lindequist, U. (2001). Screening of Yemini medical plants for antibacterial and cytotoxic activities. J. Ethnopharmacol 74:173-9. Ayyanar, M and Iganacimuthu, S. (2005). Traditional knowledge of kani tribals in Kouthalai of Tirunelveli hills. Tamil Nadu, India. Journal of Enthopharmacology 102: 246-255. Baerheim svendsen, A and Scheffer, J. (1992). Natural products in therapy. Prospects, goals and means in modern research. pharm. wkly. (sci) 4: 93-103. Baker, J.T., Borris, R.P., Carte, B et al. (1995). Natural products drug discovery and development. New perspective on international collaborations Nat prod 58: 1325-1357. Balandrin, M.F., Kjocke, A.J., Wurtele, E.S and Bollinger, W.H. (1985). Natural plant chemicals: sources of industrial and mechanical materials, Science 228-1154 -1160. Bayer, A.W., Kirby, M.D.K., Sherris, J.C and Trick, M.(1986). Antibiotic Susceptibility testing by standard single disc diffusion method, Am. J. Clinical pathol 45:493-496. Chopra, A and Doiphode, V. (2002). Ayurvedic medicine; core concept, therapeutic principles, and current relevance. med. clin. North Am 86:75-89. Dagic, E.K., Izmirli, M., Digrak, M. (2002). Kahramanmaras ilinde yetisen bazi agac turlerinin antimicrobial aktivitelerinin arastirilmasi K.S.V. Fen ve muhendislik Dergisi, Kahramanmaras 5: 38-46. Dahanaukar, S and Thatte, U. (2000). Ayurveda Revisited, Popular prakashan, Mumbai (2000) 3rd edn. Dorman, H.J.D and Deans, S.G. (2000). Antimicrobial agents from plants; Antibacterial activity off plants volatile oils. J. Applied microbial. 88: 308-316. El-Fiky, F.K., Attif, O., AboulEla, M., Gaanem, N. (1995). Antimicrobial evaluation of extract from some yemeni plants. Alex J pharm sci 9: 35-7. Evans, J.S., Pattison, E., Morris, P. (1986). Antimicrobial agents from plant cell cultures, in Secondary metabolities in plant cell culture. Morris, P., Scraggs, A., Stanfford, A., Flower, M. Cambrigde university, London P.12. Farombi, E.O. (2003). African indigenous plants with chemotherapeutic potentials and biotechnological approach to the production of bioactive prophylactic agents. African J Biotech 2: 662-671. Finar, L.L. (1983). Organic chemistry: Vol 25th Edition, pp. 696-765, Longman, London. Hamil, F.A., Apio, S., Mubiru, N.K., Bukenya-Ziruba, R., Mosanyo, M., Magangi., Ow, et al. (2003). Traditional herbal drugs of southern Uganda, II. Literature analysis and antimicrobial assays. J. Ethnopharmal 84: 57-78. Ignacimuthu, S., Sankarasivaraman, K., Kesavan. (1998). Medico-ethno botanical survey among Kanikar tribals of mundentherai sanitary fitoterapia. 69: 409-414. Krishnaraju, A.V., Roa, T.V.N., and Sundararaju. (2005). Assessment of bioactivity of Indian medicinal plants using Brine shrimp (Alternaria salania) lethality assay. Int. J. Appl. Sci Engg 2:125-134. Mothana, R.A.A and Lindequist,U. (2005). Antimicrobial activity of some medicinal plants of the island sogotra. J. Ethnopharmacol 96:177-81. Mothana, R.A.A., Mentel, R., Reiss, C., Lindequist, U. (2006). Phytochemical screening and antiviral activity of some medicinal plants of the island soqotra. Phytother Res 20:298-302. Motsei, M.L., Lindsey, K.L., Vanstaden, J., Jaeger, A.K. (2003). Screening of traditionally used South African pants for antifungal activity against Candida albicans. J Ethnopharmacol 86: 235-41. Natarajan, B., Paulsen, B.S., Pushpangadan, P. (1999). An Enthopharmacoloical study from the Coimbatore district, Tamil nadu, India. Traditional knowledge compared with modern biological science, pharmaceutical biology 37: 378-390. Patwardhan, B and Hooper, M. (1992). Ayurveda and future drug development. Int. J. Alternative complement, med 10:9-12. Purohit, S.S and Mathur, S.K. (1999). Drugs in Biotechnology Fundamentals and applications. Purohit, S.S. Maximillan publications, India P. 576. Rajan, S., Sethuraman, M., Mukherjee, P.K. (2002). Ethnobiology of the Niligries Hills, India, phytotherpy-Research 16: 98-116. Samy, R.P., Ignacimuthu, S., Sen, A. (1998). Screening of 34 Indian medicinal plants for antibacterial properties. J. Ethnopharmacol. 62: 173-81. Sandhy, B., Thomas, S., Isabel, W., Shenbagavathai, R. (2006). Ethnomedicinal plants used by the valaiyan community of Piranmalai hills (Reserved forest), Tamil Nadu, India. A pilot study, African journal of traditional complements and Alternative medicines 3:101-114. Sofowora, A. (1982). Medicinal plants and Traditional medicine in west Africa, John wiley and sons, pp. 256, New York. Stuffness, M and Douros, J. (1982). Current status of the NLT plant and animal product program. J Nat prod 45: 1-14. Suffrendini, J.B., Sader, H.S., Goncalves, A.G., Reis, A.O., Gales, H.L., Veralla, A.D., Younes, R.N. (2004). Screening of antimicrobial extract from plants native to the Brazilian Amazon rain forest and Atlantic forest Brazil .J.med.Biol.Res 37:379-384. Tan, A. (1992).Turkiyede bitkisel cesitlilik Ve bitki genetic Kaynaklari, Anadole J. AARI 2: 50-64. Trease, G.E and Evans, W.C. (1989). Pharmacognosy 11th Edn. Brailliar Tiridal Can Macmillian publishers. World Health Organization, (2002). WHO Traditional medicine strategy 2002-2005, World Health organization. Zhang, X. (2004). Traditional medicine, its importance and protection, In: Twarog, S., Kapoor, P. (eds). Protecting and promoting traditional knowledge: System, National experiences and International Dimensions .Part-I. The role of Traditional knowledge in Health care and Agriculture. New York; United nations 3-6.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||