|
Ethnobotanical Leaflets 13: 316-19. 2009.
Correlation between the Diversity Manifestation and Phytoplankton Productivity in Pechiparai Reservoir, Kanyakumari District, Tamil Nadu, India
P.J. Jepa Chandera Mohan1,2, S. Godwin Wesley1, S. Ramya2, N. Alaguchamy2, M.Kalayanasundaram2 and R. Jayakumararaj3
1 Department of Zoology, Scott Christian College, Nagercoil, KK Dist, TN, IN 2 Department of Zoology, Raja Doraisingam Government Arts College, Sivagangai 630561, IN 3 Department of Botany, RD Government Arts College, Sivagangai 630561, IN
Issued 15 February 2009
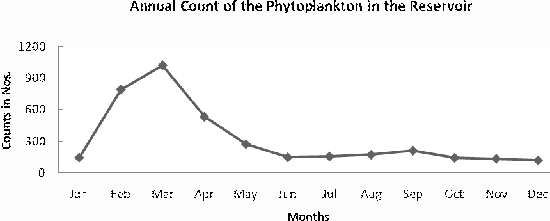
ABSTRACT Correlation between the diversity and primary production index of phytoplanktons of Pechiparai Reservoir in Kanyakumari District, TN, India was assessed from January to December 2004. Average phytoplankton primary production varied significantly during the period of study. Maximum diversity and primary productivity were record in the months of Jan to Mar. The inflow of water during the monsoon into the reservoir was the prime factor that influenced input of the nutrients and appeared to enhance the primary production during this period. Shennon Weiner diversity index varied between 1.242 and 1.897 during the period of study. Likewise, Sympson index varied from 0.822 to 0.898 and evenness from 446 to 956. The value of annual average of phytoplankton biomass was 319 Nos-1. The value of annual average of gross primary production and net primary production were 2719 mg C/m3/d and 1610 mg C/m3/d respectively. KEYWORDS Pechiparai Reservoir; Primary Production; Phytoplankton Diversity; Shannon Weiner diversity index; Sympson index. INTRODUCTION A number of factors have been attributed to influence the diversity of fauna and flora in any aquatic ecosystem. An assessment of biological productivity of any aquatic ecosystem is essential to ascertain whether the productivity is sufficient enough to support the standing stock of commercially important fishes. Topical reservoirs are generally characterized by rich population of Phytoplankton (Srinivasan, 1964). Among the factors that influence the diversity of fauna and flora of a reservoir ecosystem, phytoplankton play a pivotal role in the production of organic matter, which in tern is decided by the various environmental factors. Studies on the primary productivity in Indian lentic systems are meager (Srinivasan, 1964; Joshi et al., 1995; Sukumaran and Dhas 2001; Shrivastava, 2005). Earlier we reported the influence of spatial and temporal variations on phytoplankton community structure in Pechiparai reservoir (Jepachandramohan et al., 2009). In the present investigation correlation between the primary productivity and phytoplankton diversity in Pechiparai Reservoir in Kanayakumari District TN, India (Fig. 1) was assessed to provide the inter relationship between the primary productivity and phytoplankton diversity of reservoirs in South India. MATERIALS AND METHODS The investigation was carried out over a period of twelve months Jan 2004 to Dec 2004. Phytoplankton samples were collected by filtering the water through bolting silk cloth (No. 25) and preserved in 4% formalin. The systematic identification of the phytoplankton was done adopting the standard keys of Desikachary (1959) and Cramer (1984). The enumeration of phytoplankton count was carried out with the help of a haemocytometer. The species diversity indices, viz., Shennon Weiner diversity index (H1), Sympson index (SI) and evenness index (D) were computed. Assessment of primary productivity was done as per the procedures of Apha (1992). RESULTS AND DISCUSSION The species diversity H1 ranged from 1.242 (Sep) to 1.897 (Jan), SI from 0.614 (May) 0.898 (Jul) and J1 from 0.646 (Apr) to 956 (Aug) as shown in Table 1. The primary productivity in the planktonic algae is maximum in the upper photic zone of the water body. Therefore, assessments of the standing crop and rate of production are important factors that determine the fishery potential (Salakar and Yearagi, 2004). The seasonal abundance of phytoplankton in Pechiparai reservoir was very high during post secondary monsoon season (Jan to Mar) than the post primary monsoon season (Aug and Sep) (Fig. 2). The phytoplankton population was considerably low during period of high precipitation; this could be due the high turbid nature of the water in the reservoir which in turn brings about the decline in the intensity of light that pears though the water on the upper region (Sugunan, 1980). A direct relationship between monsoon flow and plankton density has been reported in Richand reservoir (Nataraj, 1976). Shennon Weiner diversity index (H1) and Sympson index (SI) of the reservoir increased due to varied proliferation of phytoplankton during the two post monsoon seasons. This observation is in agreement with the previous report of Sugunam (1991) for net plankton in Nagarjunasargar reservoir. Sukumaran and Karthikeyan (1999) recorded similar observations in the species diversity of peri-phyton in Markonahalli reservoir. Decline in the values of diversity indices in the southwest monsoon and northeast monsoon coincide with the decline of species index of the phytoplankton community. This observation is in accordance with that of Margalef (1963). The establishment of some species during certain months during certain months could be attributed to the prevalence of favorable environmental conditions. Evenness index j was higher in certain months apparently due to comparable distribution of various genera and the species in the habitat as affirmed Whilm and Dorris (1968). Phytoplankton synthesis new organic carbon by the process of photosynthesis in the present study the values of gross primary productivity and net primary productivity were higher in the post monsoon seasons. Subbamma (1993) observed that the population of the phytoplankton was much higher in the summer than in other seasons. This could be due to the prolonged day length and high intensity of light during this period. Srinivasan (1964) and Jepachandramohan et al., (2009) observed similar results in Ammaravathi reservoir and in Pechiparai reservoir in the earlier studies. In the present study the maximum diversity was recorded in the post monsoon period that is in the months of Jan - Mar. This could be due to the prolonged availability of light in the post monsoon period and significantly high inflow of nutrients during the monsoon. Based on the studies it is concluded that there exists a strong correlation between the population, diversity and the productivity of the phytoplanktons. ACKNOWLEDGEMENTS The authors are thankful to Principal, RDGAC and Management of Scott Christian College for their constant support and encouragements. The authors express their gratitude to Prof. G Muthiah, Head Department of Zoology, RDGAC for valuable comments and suggestions during the preparation of this manuscript. REFERENCES 1. Apha (1992). Standard method for the examination of water and waste water. 18th Ed. Washington DC. 2. Desikachary K (1959). Cyanophyta ICAR Publication. New Delhi, India. 3. Jepachandramohan PJ, Godwin Wesley S, Ramya S, Alaguchamy N, Kalyanasundaram M, and Jayakumararaj R (2009). Influence of Spatial and Temporal variations on Phytoplankton Community Structure in Pechiparai Reservoir, Kanyakumari District, TN, India Ethnobotanical Leaflets 13:249-53. 4. Joshi BD, Bisht RCS and Samual VP (1995). Primary productivity in Western Ganga cannel at Howrah Indian Journal of Ecology 21(2):123-126. 5. Margalef R (1963). On certain unifying principles in ecology. An Nat 97:357-374. 6. Nataraj VV (1976). Fish farming in man made lakes. Indian farming 26(6):24-33. 7. Salaskar PB and Yeragi SG (2004). Primary productivity in Powai lake, Mumbai, Maharashtra. Journal of Aquatic Biology.19 (1):19-22. 8. Singh DN and Das AK (2002). Spatial-temporal distribution of phytoplankton in relation to physio-chemical features in peninsular lake. Proceeding National Academy of Science, India. 72(3-4):293-303. 9. Srinivasan A (1964). Hydrological study of a tropical impoundment Bhavanisagar reservoir, Madras state, India, for the year 1950-61, Hydrobiologia 54:514-539. 10. Srivastava NP (2005). Plankton status of Ravisankarsagar reservoir. Journal of Inland fish Society, India. 37(2):43-47. 11. Subbamma DV (1993). Primary production in temple pond and a fish pond in AndraPradesh, India. Journal of Aquatic Biology 18(142):7-9. 12. Sugunan VV (1991). Changes in the phytoplankton species diversity index due to artificial impoundment in river Kroshma at Nagarjunasagar. Journal of Inland Fish Society, India. 23(1):64-74. 13. Sukumaran PK and Das AK (2001). Distribution of phytoplankton in some fresh water reservoir Karnataka. Journal of Inland Fish Society, India. 33(2):29-36. 14. Sukumaran PK and Karthikeyan M (1999). Seasonal abundance and species diversity of periphyton in Markonahalli reservoir, Karnataka Journal of Inland Fish Society, India 31(2):93-98. 15. Whilm JR and Dorris TC (1968). Biological parameters of water quality. Biosciences 18:447-481.
Table 1. Primary Productivity of Phytoplanktons in Pechiparai reservoir.
Fig.1. Location of the study area (Pechiparai Reservoir).
Fig. 2. Diversity of phytoplanktons in the Pechiparai reservoir.
|